phthalocyanine pigment
The main body in the molecule is phthalocyanine, and the structural formula is:
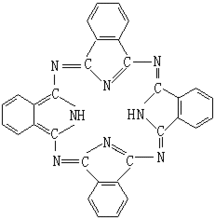
They are water-insoluble organics, mainly blue and green pigments. Most phthalocyanine pigments contain divalent metals, such as copper, nickel, iron, manganese, etc. some also replace the benzene ring in the molecule with pyrrole ring or other rings, and introduce other groups into the molecule. Different structures have different properties and uses.
The main variety of phthalocyanine pigment is copper containing phthalocyanine blue (C.I. Pigment Blue 15). In industry, the main production method is to react phthalic anhydride with urea (or phthalonitrile directly) with cuprous chloride in the presence of ammonium molybdate catalyst. The crude product is commonly known as "copper phthalocyanine".
Different products can be obtained by different methods of pigment post-treatment. For example, the crude product can be dissolved in concentrated sulfuric acid and then precipitated slowly in water α Crystal form, which is a blue organic pigment with red light; If the crude product is dissolved in concentrated sulfuric acid, and then a small amount of chlorine is introduced to make the molecule contain 1 ~ 2 chlorine atoms, the color light of the product is greener than that without chlorine; If the crude product and dry sodium chloride are ground in a ball mill, a stable green light can be obtained β Crystal products.
The crude product is heated to about 220 ° C in the melt of aluminum trichloride and sodium chloride, and chlorine is introduced into the molecule to introduce 14 ~ 16 chlorine atoms. The product is a bright green pigment, called phthalocyanine green; If a small amount of bromine is introduced, the color of the product will be more yellow and bright.


