Quinacridone pigment
The basic structure of the molecule is quinacridone pigment:
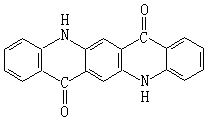
Quinacridone pigment is a kind of high-performance organic pigment whose chromatography is mainly red and purple. It was produced by DuPont company in 1958. Its production method is obtained by self condensation of Diethyl Succinate with aniline, closed-loop, refining and oxidation γ Crystalline quinacridone pigment. Because its heat resistance, sun resistance and brightness are equivalent to those of phthalocyanine pigments, the commodity is called phthalocyanine red (C.I. Pigment Violet 19). In fact, their molecular structures are completely different. If different conditions are adopted during oxidation, the color light is bluer β Crystalline quinacridone pigment, commercially known as phthalocyanine violet.
The two representative products of quinacridone pigment are pigment red 122 and pigment purple 19, which are known as complementary colors relative to the blue and green chromatographic range of phthalocyanine pigment. They have bright colors, high color intensity, excellent heat resistance, solvent resistance, light and weather resistance, and are widely used in automobile coatings, high-grade industrial coatings, plastic products Coloring of metal printing ink and building materials.
At present, the research focus of organic pigment workers is on the improvement of pigment post-treatment, such as selecting better crystal form, manufacturing finer particles with narrow particle size distribution, improving the wettability of pigments, so that organic pigments can play a greater role.


